The following content has been reviewed by a licensed attorney.
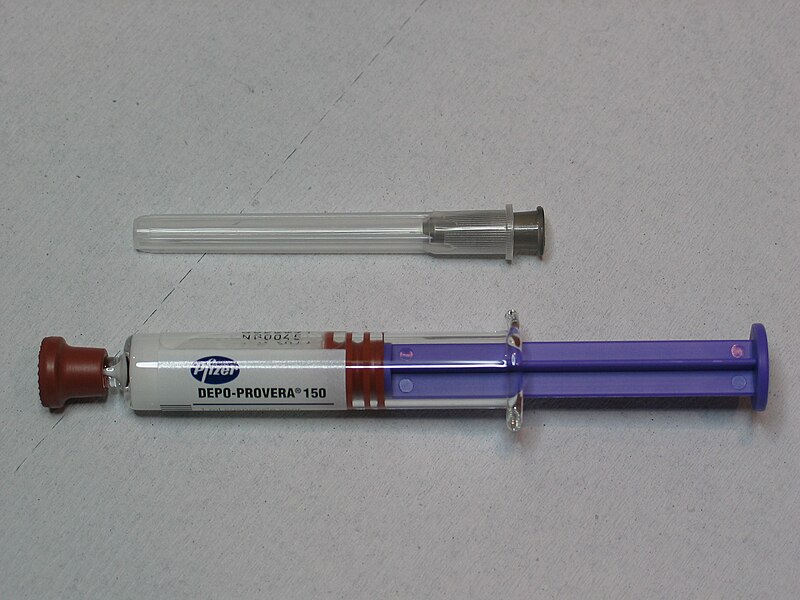
Depo-Provera, a widely used injectable contraceptive, has come under intense scrutiny following alarming revelations about its safety. A 2024 scientific study linking the use of Depo-Provera to an increased risk of meningiomas, a type of brain tumor, has sparked a wave of litigation. These legal challenges contend that Pfizer, the pharmaceutical giant behind Depo-Provera, had prior knowledge of these risks and failed to warn the public adequately.
Overview of Depo-Provera
Depo-Provera is an injectable form of birth control that contains medroxyprogesterone acetate, a synthetic hormone similar to progesterone. It is administered every three months and is favored for its efficacy and convenience. However, despite its popularity, Depo-Provera has been associated with a range of adverse effects, including changes in menstrual cycles, weight gain, and decreased bone density. The recent findings regarding its link to meningiomas, however, represent a far more severe and concerning potential side effect.
The Scientific Study and Its Findings
The pivotal study that brought these issues to light was published in a leading medical journal and drew upon extensive patient data. It found that the risk of developing meningiomas was significantly higher in women who used Depo-Provera, especially those who used it long-term. Meningiomas, while often benign, can grow large enough to cause serious neurological complications, necessitating surgery and other invasive treatments.
This research suggested a dose-response relationship, indicating that the longer the exposure to Depo-Provera, the greater the risk of developing these tumors. The implications of these findings are particularly distressing given the number of women who have relied on this medication for their reproductive health needs.
Legal Implications and Pfizer’s Accountability
In response to the study, numerous lawsuits have been filed across the United States, alleging that Pfizer had knowledge of Depo-Provera’s potential to induce meningiomas but did not provide adequate warnings. These lawsuits claim that Pfizer’s failure to inform has violated consumer trust and endangered health.
Documents unearthed during legal proceedings suggest that Pfizer might have been aware of the potential link between Depo-Provera and brain tumors from earlier studies and internal research. If proven, this knowledge underscores a significant ethical lapse in Pfizer’s responsibility to its consumers and raises questions about the regulatory oversight of drug safety.
The Push for Multi-District Litigation (MDL)
Given the growing number of lawsuits, there is a strong push to consolidate these cases into a Multi-District Litigation (MDL). This would streamline the management of pretrial proceedings, including discovery and evidentiary admissions, which are common to the cases. An MDL would not only expedite the handling of these numerous and complex cases but also potentially lead to a more uniform resolution.
Broader Implications for Pharmaceutical Practices
The Depo-Provera controversy highlights critical issues in pharmaceutical regulation and consumer safety. It underscores the need for more stringent post-marketing surveillance of medications and better transparency in how side effects and risks are communicated to the public. This case could serve as a catalyst for regulatory reform, emphasizing the need for pharmaceutical companies to maintain rigorous safety standards and for regulators to enforce stringent oversight.
Future Directions and Consumer Impact
As the legal proceedings advance, they will likely influence how drugs are regulated and how pharmaceutical companies approach the disclosure of drug-related risks. For consumers, this case serves as a reminder of the importance of being informed about the medications they use and advocating for their rights to comprehensive and honest information.
The outcome of the Depo-Provera litigation will have far-reaching consequences not only for those directly affected but also for future regulatory policies and the overall approach to drug safety monitoring. It is a pivotal moment that could reshape industry standards and reinforce the imperative of prioritizing patient safety in pharmaceutical practices.
Final Thoughts
The unfolding Depo-Provera saga is more than a legal battle; it is a public health concern and a test of corporate ethics in the pharmaceutical industry. As affected individuals seek justice through the courts, the broader implications for drug safety, corporate responsibility, and regulatory adequacy remain at the forefront of public discourse. This case may well be a watershed moment in the ongoing effort to ensure that drug companies not only provide effective solutions but also conduct their business with transparency and integrity for the sake of public health.